The pain puzzle: Discussing the contributors to pain experience
By Dr. Alejandro Elorriaga Claraco
Features Clinical TechniquesUnderstanding neuroreality in pain with movement disorders
 The neurofunctional contributors to pain experience interact like pieces of a puzzle. Photo: Fotolia
The neurofunctional contributors to pain experience interact like pieces of a puzzle. Photo: FotoliaIn my previous article (April 2018) I presented and discussed the concept of “neuroreality” as the most accurate science-based conceptual foundation available to approach the understanding of “pain with movement” disorders.
In this article, I discussed that there is no objective reality and it is the brain – our brain – that creates the only reality we have access to: our neurological reality, our unique experience of the world that is only available to each of us.
The main conclusion to the article was that “current scientific evidence precludes pain models based mostly on musculoskeletal structural changes,” i.e. joints, muscles, tendons, and bones. This statement may sound categorical but, with all we currently know about neurofunction, there are enough scientific facts that explain why structures could not be the main cause of pain with movement problems. The first level of evidence is the fact that many individuals with structural lesions don’t exhibit any discomfort. These findings have been published in the peer-reviewed literature for some time, e.g. “Systematic Literature Review of Imaging Features of Spinal Degeneration in Asymptomatic Populations,” published in the American Journal of Neuroradiology (April 2015).
The rather astonishing findings included the following:
- The prevalence of disk degeneration in asymptomatic individuals increased from 37 per cent of 20-year-old individuals to 96 per cent of 80-year-old individuals
- Disk bulge prevalence increased from 30 per cent of those 20 years of age to 84 per cent of those 80 years of age
- Disk protrusion prevalence increased from 29 per cent of those 20 years of age to 43 per cent of those 80 years of age
- The prevalence of annular fissure increased from 19 per cent of those 20 years of age to 29 per cent of those 80 years of age.
These findings fully contradict the common clinical practice in back pain syndromes, of allocating blame to the structures of the back after finding something wrong after imaging. Obviously, the inevitable intelligent conclusion of the authors of the study was that “imaging findings must be interpreted in the context of the patient’s clinical condition,” they just never explained how to do that.
I’m going to present some of the elements of the neurofunctional operating system in order to help clinicians better interpret both symptoms and structural findings in pain with movement disorders, in a science-based manner. However, this task would require several hundreds of hours of continuing education working together, and cannot be accomplished in the limited space provided by the pages of this magazine. Therefore, these articles will focus in a simplified manner on some of the most important elements of the neurofunctional operating system, necessary to accurately navigate the clinical complexities of pain with movement disorders.
The neurofunctional operating system is a science-based methodology to assess and treat individuals complaining of pain with movement. Its foundation is the contemporary neurofunctional model of pain.
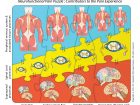
Pieces of a puzzle
Figure 1 visually presents the three main neurofunctional contributors to a pain experience (peripheral nervous system processes, spinal cord processes, and supraspinal processes), all interacting with each other like the pieces of a puzzle. It shows different sizes representing different magnitudes of contribution to the pain experience in different clinical scenarios. These clinical scenarios can be read at the bottom of the illustration and, over each of them, the vertical interaction of the pieces belonging to each group of processes give us a quantitative visual impression of their relative contribution to each clinical situation. Starting on the left, we have acute pain; a particular situation in which most of the pain experienced by the person’s brain is dependent on peripheral nociceptive activity (particularly after trauma), which is represented by the biggest piece of the whole puzzle. Acute pain follows a predictable pattern, when the peripheral nociceptive activity subsides, so does the pain. Unfortunately, in clinical practice only a few cases fall into this clear-cut acute pain scenario.
It’s more common to deal with the second type of pain problem: insidious onset pain. Now, there is no history of trauma and explanations become less scientific. Still many practitioners continue to blame the musculoskeletal tissues for the pain by using “structurally related words” like “wear and tear” or “degenerative joint disease” or “tendinopathy” among many others. However, the scientific fact remains that the pain experienced in these common cases of insidious onset pain is more dependent upon processes that occur at the spinal cord (a.k.a. spinal sensitization) and processes in the brain and associated supraspinal neurostructures (a.k.a. central sensitization), than in the peripheral tissues (even accounting for the phenomenon of peripheral sensitization often present). Since, in most of these cases, the clinical findings do not seem enough to justify the magnitude and persistence of the symptoms, what is causing this abnormal behavior of the nervous system? It seems that at the core of how most of these clinical situations arise is the phenomenon of neurogenic inflammation, which is triggered by the secretion of neuropeptides produced by the irritated nociceptive fibres (C fibres) of a given segment, which in turn produces biochemical and vasculomotor changes in the receptor fields at the peripheral tissues and in the spinal cord. This initiates a multilevel phenomenon of “sensitization” (peripheral, spinal, and central), which is at the core of most insidious onset as well as lasting pain problems.
The most costly of all scenarios
For reasons of complexity and lack of current understanding, we will leave out the discussion about some of the worst pain neuropathies such as the complex regional pain syndromes, for another occasion, and proceed to discuss the right side of the spectrum of the pain puzzle, the most costly of all scenarios, both for sufferers and society alike is chronic pain.
On this side of the spectrum, we can face different clinical situations, but all are characterized by a major contribution of the supraspinal processes to the pain experience, and not limited to the sensory dimension of pain. It also involves other negative effects systemically, as well as on the nervous system, the endocrine and immune systems, and the psycho-emotional well-being of the chronic pain sufferer. To this widespread dysfunction, we can add the side effects of medication and other interventions (a.k.a. iatrogenia), and dealing with these cases becomes uncontestable.
The situation on this side of the puzzle is very different than what it was with acute pain – we can even have a pain situation where no detectable nociceptive activity in the periphery exists, and despite this the brain experiences significant pain. It could be a case of phantom limb pain syndrome, which is even possible in cases where the person was born without the sore limb. Thalamic pain syndrome could also be placed in this same situation where all the pain symptomatology is generated by the central nervous system, generally after a stroke, without participation of any musculoskeletal structure. This is a devastating and largely intractable type of pain.
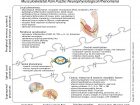
Physiological occurrences
To better understand the many neurophysiological and neuropathological events associated with the different case scenarios we have briefly discussed above, let’s take a look at Figure 2. The illustration presents a list of the main physiological occurrences that may happen at the three levels of the main neurofunctional contributors to a pain experience. Most of the time, this pain experience is a multidimensional phenomenon that not only requires the presence of neurodysfunction, but also needs additional contributions from the metabolic, biomechanical and behavioral levels. In turn, each of these areas represents another major area of study in their own right.
Treating what is ‘really’ wrong
Now, despite having a better understanding of the neurofunctional contributors to the pain experience, we still need to localize in order to be able to treat, which is the beginning of all our clinical needs. We need to understand the dysfunction and to select the best interventions available to restore the lost adaptability. To accomplish this, we need to treat what is really wrong, and what is really wrong is not the structure as we already have established. What is really wrong in a “pain with movement” disorder is neurofunction, particularly regarding connectivity among the central neurons and the peripheral receptor sites, both for the sensory, the neuromotor, and the peripheral vacular sympathetic levels, as well as the physiology of nerve transmission and the phenomena of central modulation and integration of neurosignals.
We need to map the dysfunction in a pain with movement disorder by understanding the many levels of neurofunctional organization of the nervous system, specifically peripheral nerves and spinal segments at dermatomes, myotomes and sclerotomes, including which divisions (anterior or posterior), what vertebral levels and what viscera are associated with those spinal segments.
Neurofunctional diagnosis is an ambitious clinical approach, but is the best we have in order to help generate the most effective therapeutic interventions available for the millions of sufferers with these pain with movement disorders.
DR. ALEJANDRO ELORRIAGA CLARACO is an international sports medicine consultant who has worked with hundreds of professional athletes and thousands of clients for over three decades. He has used his extensive clinical experience and research to become an innovative educator in the field of “pain with movement” disorders. You can find out more at mcmasteracupucture.com.
Print this page